bioMérieux partners with NecstGen
bioMérieux and NecstGen, collaborate to accelerate the development of Cell and Gene Therapies (CGT) through the provision of ‘state of the art’ and rapid quality and safety testing technologies that will allow safe and effective advanced therapeutics production.
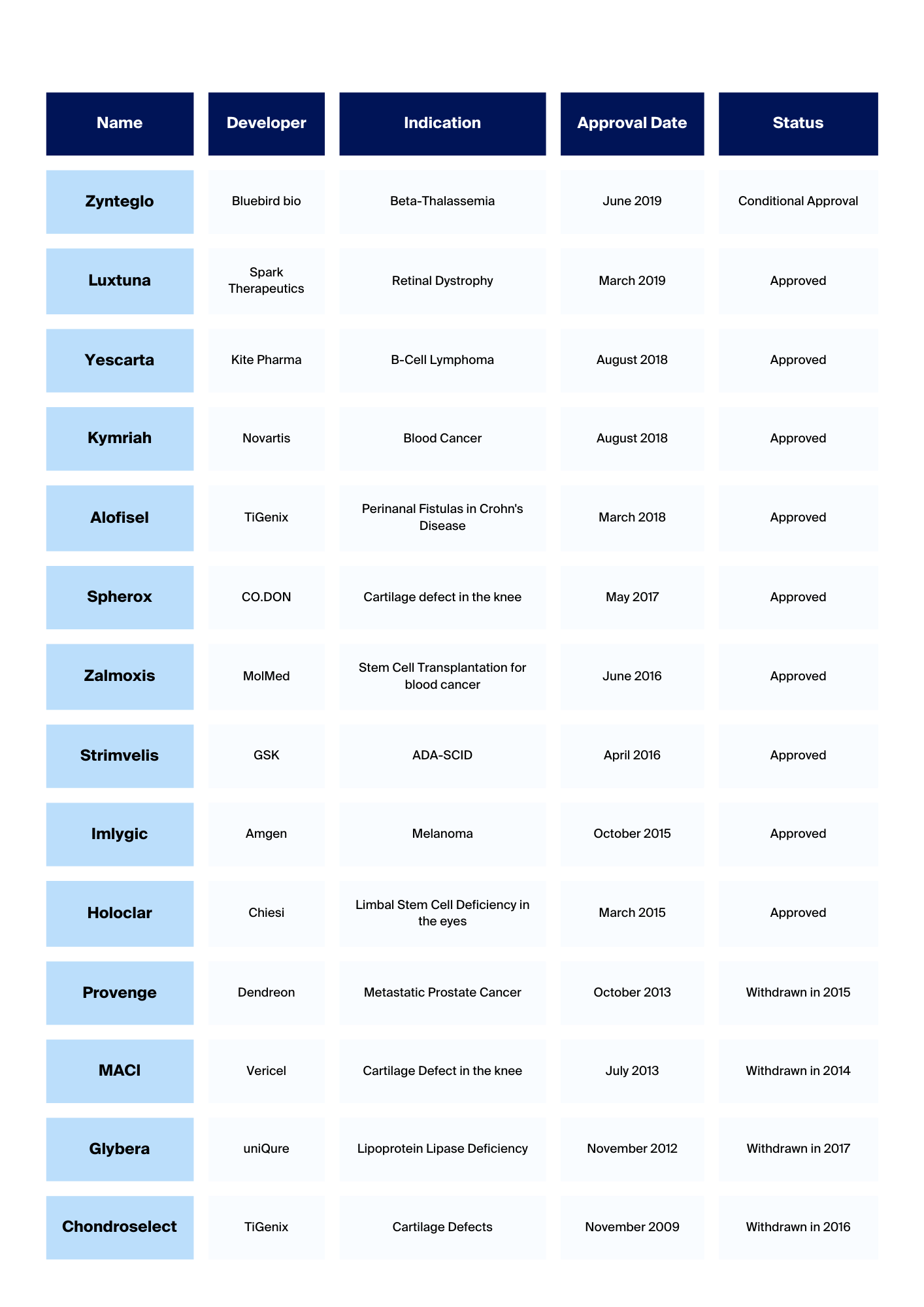
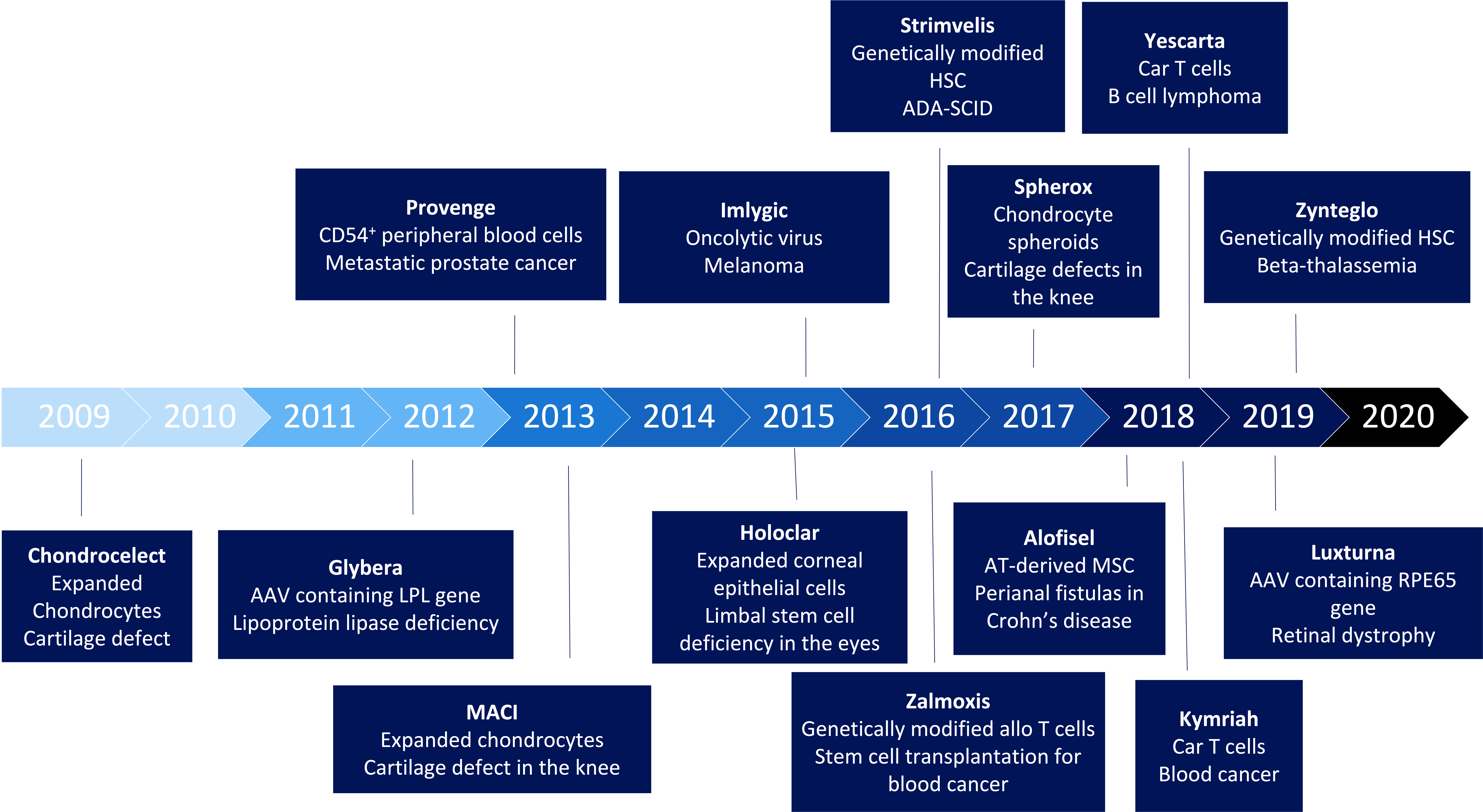
Cell and Gene therapies are revolutionary treatments centered on using patient or donor cells, cultivated or modified outside of the body and re-introduced to the patient to induce the desired therapeutic effect. This complex field of therapies has opened the door to long-term cures for diseases such as cancers and orphan diseases and offers hope to patients and clinicians alike. However, the development and manufacture of safe and effective treatments, that can reach large numbers of patients, present many challenges. Scientist must ensure not only that their advanced therapy is effective it must also be manufactured in accordance with the requirements of the regulatory bodies and free off microbial contamination. They look to innovative microbiology companies, like bioMérieux, to provides solutions that will enable them to release safe medicines onto the market.
Thanks to this new partnership, CGT researchers and manufacturers, hosted at NecstGen, will benefit from the solutions and services provided by bioMérieux to monitor and test their products and processes. bioMérieux rapid methods for Sterility testing, Mycoplasma detection and Endotoxin testing that form part of the partnership will allow them to realize good manufacturing practice (GMP) compliance and to overcome constraints specific to advanced therapies including product sacrifice for sampling, complexity of sample composition, and the need to reduce vein to vein time. Breaking down the testing bottlenecks, will help accelerate CGT development and improve access to these much-needed medicines for patients.
Christie Downing-Kay, Head of Marketing for Northern Europe Cluster, bioMérieux comments, “CGT’s are the third horizon of medicine and have the potential to cure conditions, such as ‘orphan’ diseases, previously deemed incurable. bioMérieux is passionate about supporting the development of CGT’s and believe this collaboration with NecstGen will provide synergies that will benefit this promising field and help bring medicines to the patients that need them”.
NecstGen, is a centre of excellence for CGT, providing critical contract development, manufacturing and rental services to therapy developers to deliver a new generation of therapies to patients., NecstGen is located in a purpose-built GMP facility on the largest bio-cluster in the Netherlands, Leiden Bio Science Park.
Paul Bilars, CEO at NecstGen added “We are very happy to engage in this collaboration and share knowledge in CGT manufacturing and Quality Control. Ultimately, this partnership will help us to offer our clients the best support and access to state-of-the-art systems to advance the development of their next generation therapies.”
This collaboration with NecstGen is the culmination of developing solutions adapted to the constraints of this market, as well as educational content to support the key players in the production of CGT as underlined by Michaël Reynier (Sr Vice President for the Pharma Quality Control Business, bioMérieux): “We are very pleased to join forces with NecstGen to offer CGT developers a strong portfolio of value-added Quality Control solutions within a state-of-the-art Cell Therapy development and GMP manufacturing environment to speed up path to the market and release of life saving advanced therapies.”
Overcoming complex manufacturing challenges and ensuring the quality, safety, and efficacy of cell and gene therapies administered to patients is critical. That’s why bioMérieux delivers a proven portfolio of value-added solutions to add rapid and easy-to-use technologies to operations and bring overall efficiencies to the manufacturing process through both simplified workflows and accurate, actionable results to ensure patient safety.